
Chapter 5
Non-melanoma skin cancer (NMSC)
Skin cancer can be divided into two principal types on the basis of clinical and biological differences:
- Non-melanoma skin cancer (NMSC)
- Melanoma (also referred to as ‘malignant melanoma’ (MM))
Non-Melanoma Skin Cancer (NMSC)
These types of cancer are derived from keratinocytes and are far more common than melanoma, but have a much lower case-fatality (<0.1%). The principal types of NMSC are:
- Basal cell carcinoma (BCC)
- Squamous cell carcinoma (SCC)
There are in addition a number of other lesions that are pre-malignant or related to skin cancer.
- Actinic keratoses (AK)
- Bowen’s disease or intraepithelial carcinoma (IEC)
- Keratoacanthoma (KA)
So, at least initially, you have to think about 5 conditions, 2 are cancer proper and the other 3
are premalignant or related in some other way to either BCC or SCC.
Actinic keratoses —premalignant lesions
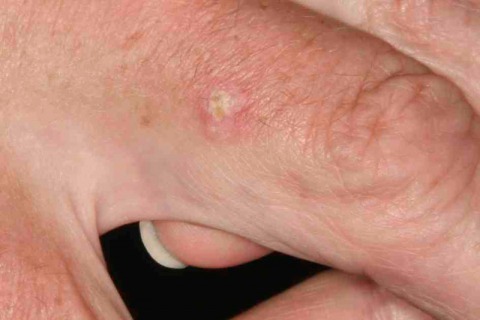
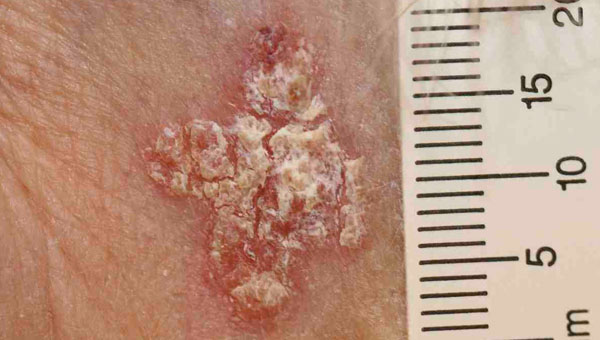
Actinic keratoses are focal areas of dysplasia characterised by erythema and scale that usually feel rough when the fingers are rubbed over them [1]. They are a marker of UVR exposure occurring at those sites that have received the most cumulative sun exposure such as the scalps of bald-headed men, the backs of the hands and the nose and ears. They are most common in those with sun sensitive skin and in those with high UVR exposure (and hence increase with age). They can be considered precursors of squamous cell carcinoma in that a very small proportion of actinic keratoses progress to squamous cell carcinoma. AKs vary in size from 1mm to over 1cm in diameter. They are frequently multiple with some patients having over 20 lesions or more.
The distinction between sun damaged skin and early AKs may be not be straightforward, as in many patients with multiple AKs, you can see or feel small areas of erythema and scale that if biopsied would show dysplasia. There is considerable clinical variation in what experienced clinicians will describe as an AK. Exact figures for AK incidence are therefore not surprisingly not known, but clinically they are the most common dysplastic or neoplastic lesion of skin. The author can rarely sit on a bus in Edinburgh without spotting many AKs on other elderly passengers.
Actinic keratoses are focal areas of dysplasia which are typically red and scaly.
Should all AKs be treated?
The exact proportion of AKs that progress to SCC is still debated and this influences views on how aggressively they should be sought out and treated. For instance one estimate is that the chance of an AK progressing to an SCC is around 1:1000/year. It is also thought that many regress (at least over the short term)—perhaps around 25% in one year. It is therefore possible to argue that (some) AKs may not need treatment, but rather that effort should be focussed on early detection of SCC. There is however little or no systematic evidence on which to base strong recommendations. In practice the default option is to treat AKs with the following arguments being used to justify this course of action.
- Cosmesis or appearance. Most people would prefer not to have a heaped up scaly red lesion on their skin.
- Some AKs may progress to SCC and we have little explicit criteria to decide on which ones will regress or fail to progress.
- You cannot always clinically be certain that a larger AK is not an IEC or SCC.
My own view is that there may be considerable heterogeneity in AK behaviour related to their size with larger lesions being more worthy of attention. Similarly, I doubt there is a clear distinction between small AKs and the histological abnormalities frequently seen in sun damaged skin without clinical evidence of an AK.
Treatment of actinic keratoses
AKs are superficial lesions and the abnormal clones of cells are close to the skin’s surface (within the epidermis). Excision is not therefore usually required. THe most consistent and traditionally widely used treatments are:
- Cryotherapy — application of liquid nitrogen to destroy the superficial area of skin
- Curettage and cautery (“C +C”) — scraping of the skin surface under local
anaesthesia
Given the frequent occurrence of a large number of lesions cryotherapy is often used as the first line treatment. Further details of these treatments and others such as topical chemotherapy that may be used to treat AKs are described in the next chapter on treatment of NMSC. Finally, if the clinical diagnosis is assured then histological confirmation is not normally required [2]. However if samples are taken (as in curettage and cautery) it is advised that such samples are sent for histopathological confirmation. Inevitably some AKS will be excised because they were thought to be SCCs clinically.
- This is one of those instances where tactile information can provide informations
that is often not so apparent to one’s eyes. Clinically you often see one AK and
then rub your hands over another area of skin and pick up other less visually
obvious lesions. - What about the mantra of ‘sending all specimens for pathology’? As a general rule,
if you remove any skin lesion (whether by excision or curettage) you should send it
for histopathological examination. Obviously, if you treat a lesion with cryotherapy,
then you will usually not have obtained prior histopathological confirmation of the
diagnosis. This is a situation whereby you have to be confident of your skills as
distinguishing AKs from early BCCs is not always easy: if in doubt perform a biopsy
before cryotherapy.
Intraepithelial carcinoma (IEC, Bowen’s disease)—premalignant lesions
Intraepithelial carcinoma is in situ carcinoma of the skin. That is, the clone of cells is confined to the epidermis and has not breached the basement membrane. By contrast with actinic keratoses, in IEC the dysplasia involves the full thickness of the epidermis. One can envisage IEC as further along the pathway of carcinogenesis than an AK, and the rate of progression to SCC from IEC is judged to be much higher at around 5-10%than for AKs [1].
IEC is the result of sun exposure and therefore follows the usual risk factors. However some IEC may be the result of other carcinogenic agents such as arsenic. Histopathological confirmation of the diagnosis may be required with an incisional biopsy (that is you do not excise all the lesion, but just take a sample for histology) to assay for any invasion (and hence the presence of an SCC). All surgical samples of presumed IEC must be sent for histopathological examination. We do not have accurate incidence figures for IEC but they would appear to be less common than AKs but more common than BCCs.
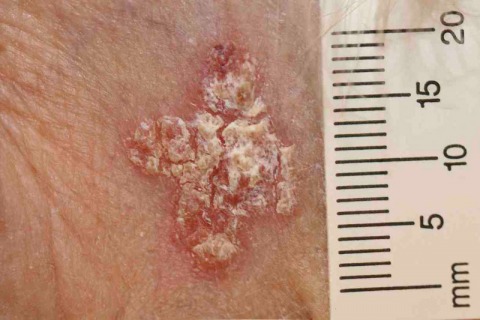
The use of the term IEC and Bowen’s disease is confusing. Some AKs show full thickness dysplasia and therefore are by the definition given above are IEC (‘Bowenoid actinic keratosis’). However the term Bowen’s disease is often clinically used to refer to an area of dysplasia that has a characteristic clinical picture that resembles a plaque of psoriasis and is found on sites such as the trunk or the lower legs, most commonly in elderly women. These lesions may be quite large, up to 6cm or more, and are often well defined plaques. Although these plaques are much more common in those with greater sun exposure, it is likely that some other cofactor must play a role in their pathogenesis as their appearance and focus on the lower limbs is distinct.
IEC is full thickness dysplasia of the epidermis. Clinically it is often reminiscent of a patch of psoriasis or discoid eczema. On other occasions IEC resembles a larger actinic keratosis.
Treatment of Bowen’s disease
The usual therapeutic options are either
- Cryotherapy
- Curettage and cautery (C+C)
- Excision.
- Topical chemotherapy
In the UK this is probably the order of preference of therapy. More advice on management of Bowen’s disease is given in the next chapter.
- Astute readers will (rightly) be disturbed by statements that the rate of progression from IEC to SCC is around 5-10% (or any other figure for that matter). Over what time period does this 10% refer to? In reality we have little robust data to go on beyond a lot of clinical experience and the not infrequent occurrence of lesions which from history have been IEC and then which appear to show progression to SCC. The best I can say is that these figures are widely used to guide clinical decisions as a sort of ranking of clinical risk.
Basal cell carcinoma (BCC)
Basal cell carcinomas are keratinocyte tumours that histologically resemble basal cells but the exact keratinocyte subset that gives rise to them is not known [1]. There are no known precursors of BCC. AKs and IEC are not precursors of BCC, but of course people who have had AKs or IEC are at increased risk of BCC because all these diseases relate to prior UVR exposure.
Vignette
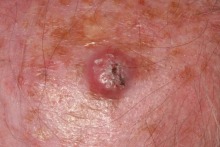
The typical BCC measures less than 1 cm across, has a pearly edge surrounding a small ulcer and shows prominent telangiectasia, and is located on the middle third of the face. They often present as lesions that start off very small and are noticed by the patient to be a cut that doesn’t heal before steadily growing larger. They may appear over a matter of months or be present for many years growing only very slowly.
Note the pearly edge, the telangiectasia and the ulceration. Not all BCCs are so stereotypical.
Epidemiology
BCCs are the commonest human cancers in those with pale skin with an incidence of approximately 100/100,000 in Northern Europe. The median age of cases is 67, and the sex incidence is equal. BCC incidence increases with age, and they are much more common in those with pale skin who have lived in areas with high ambient UVR.
BCC are most common on the middle third of the face, but are found on other sun exposed sites too such as the scalp, back, legs and arms. Very (very) rarely they occur on sun protected sites such as the genitalia or buttocks. BCCs on less frequently affected sites are often harder to diagnose as they tend to look atypical.
The majority of patients who present with BCC will have no risk factors beyond being relatively pale skinned and exposure to UVR. There are however certain additional risk groups.
- BCC are more common in those receiving immunosuppressants following organ
transplantation (as are the other types of NMSC) - There are rare inherited syndromes that feature BCC. The most noted of these is Gorlin’s syndrome also known as the Nevoid Basal Cell Carcinoma Syndrome . This syndrome is inherited as an autosomal dominant and is due to a mutation in a tumour suppressor gene called patched gene that plays a key role in epidermal physiology. Clinically these patients present with a large numbers of BCCs at a young age and show small pit-like abnormalities on their palms as as well a variety of extracutaneous abnormalities, and dysmorphic facies. There may not be any family history as new mutations are common and expressivity varies. Our understanding is that the first ‘hit’ is inherited and the second is the result of UVR.
- X -irradiation or other forms of ionising radiation. Hard though it may now be to believe, in the past X-irradiation was used for a number of non-cancer skin disorders such as ringworm (dermatophyte infection) or congenital vascular anomalies. Such treated areas are at an increased risk of BCC. One special case here are patients with Gorlin’s syndrome who should not be treated with radiotherapy (for their BCCs or any other reason) because they respond with an even greater number of BCCs in the areas that are irradiated [2].
- The history of a previous BCC or other type of skin cancer increases the risk of a further tumour. It is not at all uncommon to diagnose more than one BCC on the patient at the same time, and it is for reason that the clinical mantra is always: “you must undress a potential skin cancer patient adequately”.
BCC Behaviour: invade but ‘never’ metastasise
Basal cell carcinomas are unique in the sense that whereas they are invasive they virtually never metastasise [3]. Although metastasis is so rare, that they are invasive still means that not only can they invade the dermis but they can grow down and (for instance) invade cartilage, or the orbit, and track down nerves such as the supraorbital nerve. Their familiarity to doctors should not hide the fact that in a minority of cases cause significant morbidity and, very rarely, even death.
Not all BCC look the same
Dermatology would be much easier to learn if all instances of a particular diagnosis looked the same, but this is not the case. Remember not all patients with ischaemic heart disease present the same way either. For BCC whilst there may be a continuum of appearances there are various clinical pathological subtypes that are clinically useful, and influence management. Some examples include:
- Nodular BCC: These are usually the classical BCC that are often shown in textbooks.
They are often well defined and can be diagnosed confidently and excised with a lot
of confidence as to where the tumour margins are. - Morphoeic BCC: Defining the edge of these tumours is subject to considerable error,
and they can sometimes be several centimetres larger than they first appear. This
means that defining the edge to allow excision with a 4mm margin is inadequate and
Mohs’ surgery would appear more appropriate (Mohs’ surgery is discussed in the
next chapter). - Superficial BCC: These are relatively more common on the back and limbs. They do
not show any induration and non-surgical approaches may be more appropriate than
excision for some of these cancers.
Contrast these two lesions with the nodular type BCCs in Figure 5.3 — at the level of physical appearance they have little in common
Treatment of BCC
Confirmatory histopathology is required in virtually all cases. BCC cannot always be reliably distinguished from a number of related tumours (especially appendageal tumours [4]). The treatment of choice is excision with a 4mm margin. There are however some caveats to add to this simple statement, as some may be treated with curettage or even cryotherapy. Therapy is discussed in detail in the following chapter.
- BCC appear to be made up of cells that resemble basal cells histologically—this does not mean that they are derived from basal cells
- Remember in a person with Gorlin’s syndrome every cell in the skin (and body) already has ‘one hit’ on the way to cancer. It is therefore not too surprising that a mutagen like X-irradiation can supply a second hit and accelerate the progression to clinical BCC
- Never say ‘never’ is a time honoured medical heuristic. Perhaps 1:4000 BCCs does metastasise and usually these are very large and neglected tumours. In practice >99.9% of tumours are managed as thought they are incapable of metastasis.
- Appendageal tumours are tumours—either benign or malignant—of the hair follicle, or eccrine or sebaceous gland. There are a myriad of such types of tumours and they often look clinically like BCCs. Remember that one theory maintains that BCC are themselves hair follicle derived, so it is perhaps not too surprising that they can be confused clinically (and even pathologically).
Skin
Squamous cell carcinoma (SCC)
Squamous cell carcinomas are keratinocyte tumours that appear on histological grounds to be more differentiated than BCC in they appear histologically nearer to terminally differentiated keratinocytes than basal cells. It is a frequent mistake to believe that this means that they are derived from the terminally differentiated cells above the proliferative compartment in the epidermis—this cannot be the case, as these cells being terminally differentiated, have lost the ability to divide and therefore cannot give rise to malignant clone. As with so much dermatology, nomenclature reflects morphology rather than ontogeny.
Clinical Vignette
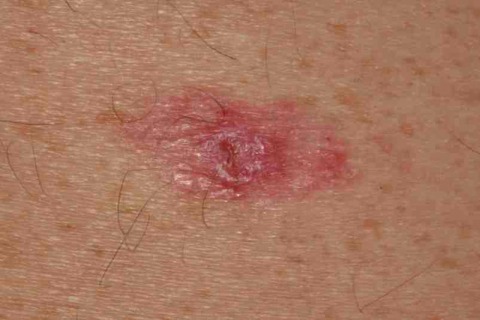
The typical SCC is a keratinising nodule that may be ulcerated or show excess keratin formation. They are often referred to as ‘ugly’ and ‘untidy’ looking. Given their relation with high levels of cumulative UVR exposure the surrounding skin may show many features of photodamage including dermal atrophy, dyspigmentation, and an increased variations in skin vasculature. They vary in size from ~5mm up to 6cm or greater. Some poorly differentiated SCCs appear to have lost their ‘keratinising’ appearance and resemble a piece of raw steak or jam on the skin’s surface.
Note the extensive photodamage especially for the image on the right. The ulceration is far more extensive on the lesion of the left.
Epidemiology
SCC are less common than BCC with an incidence of around 20:100,00 in Northern Europe, with a median age of 75. They are more common in men than women (3:2). SCC are most common on the sites that receive the largest amount of cumulative UVR such as the tops of the ears, face and back of hands. Their body site distribution therefore is identical to that of AKs, which are precursors of SCC, but different from BCCs and, as we shall learn, different from melanomas.
Most patients will have no risk factors except relatively pale skin and sun exposure. There are however a number of other risk factors:
- Immunosuppression: SCC are dramatically more common in those patients with organ transplants and who are therefore receiving immunosuppressives. Because immunosuppression itself may allow a tumour to behave more aggressively and SCC (unlike BCC) metastasise, SCC represent a significant mortality risk to this patient group. Management of such patients requires regular expert surveillance of the skin, advice on minimising UVR exposure and prompt surgical excision of any SCC that do develop. In such patients both the clinical features and histopathological appearance of the tumours is more complex than in immunocompetent individuals and more caution in management is warranted (i.e. waver on the side of over rather than under-treatment).
- Inherited syndromes: There is no inherited tumour suppressor syndrome as there is for malignant melanoma or basal cell carcinoma. SCC are however a feature of xeroderma pigmentosum (along with increased numbers of BCC and melanomas).
- Other carcinogens. In areas with high levels of arsenic ingestion cutaneous SCC (as well as SCC of other internal organs) is more common. Patients who have received large amounts of PUVA (Psoralen+UVA) therapy especially when the PUVA therapy was used on a regular basis over long periods are much more prone to SCC development. The main group of patients affected is a cohort of patients from the 1980s and early 1990s when PUVA was — with hindsight — overused for the treatment of plaque psoriasis.
Clinical behaviour
SCC are both invasive and capable of metastasising and therefore of causing death. As a general rule SCCs of sun exposed skin are less aggressive than SCCs of other keratinocyte derived tumours such as the SCC of the cervix or mouth or oesophagus. The reason for this is unclear. Actinic keratoses and Bowen’s disease are precursors of SCC.
Treatment of SCC
SCC should be excised with a margin of normal tissue of 4-6 mm. This might mean that flaps or grafts are required (discussed in the next chapter)
Keratoacanthomas
Keratoacanthomas are biologically fascinating lesions in that they look like SCC both clinically and histologically, but their behaviour is radically different — after a period of rapid growth they regress leaving an unsightly scar [1]. Their clinical importance is therefore that they are easily confused with a fast growing SCC, so much so, that many of us believe they cannot be reliably distinguished on clinical appearance alone, and instead should be treated as SCCs until proved otherwise.
Clinical Vignette
Keratoacanthomas tend to grow very quickly over a period of 6-8 weeks attaining a size of 2-6cm or even larger. They resemble a volcano with the central plug comprising keratin within a symmetrical and smooth cylindrical shoulder. They are most common on sun exposed sites. However, unlike SCC they involute without any treatment. They do not metastasise. When they do disappear they tend to leave an unsightly scar.
Usually described as having a volcano like structure, with the‘volcanic plug’ being made of keratin. Often it is not possible to distinguish a KA from an SCC. Of these two lesions the one on the right could easily be a fast growing SCC.
We do not have accurate incidence figures for KAs but they are less common than SCCs and are more common in the usual high risk groups (sun sensitive skin and high UVR exposure).From a clinical (rather than biological) perspective the main issue is distinguishing keratoacanthomas from SCCs. The short rapid growth is a clue as is the typical ‘volcano’ morphology. However, both these features can be seen in SCC which are more common than keratoacanthomas; the problem is that not treating a SCC is a worse offence that overtreating a keratoacanthoma. In practice the fact that keratoacanthoma heal leaving an unsightly scar suggests that keratoacanthoma should be excised as though they were SCC. This approach is bolstered by the fact that for a histopathologist to diagnose a keratoacanthoma with confidence, an incisional biopsy needs to be across the whole lesion (rather than say an incisional punch biopsy). In the majority of instances the appropriate management is therefore to manage as for SCC with excision.
- One obvious question is whether KAs are really neoplasms or clonal at all. The available data is sparse but it appears that mutations and chromosomal loss are rare in KAs when compared with SCC. One theory of their pathogenesis is that their cyclical growth pattern reflects in some way an aberrant manifestation of the cyclical growth pattern that occurs in the hair cycle where a pattern of growth (anagen) is followed by a period of involution (catagen).
- Which is more common, BCC, SCC or AK?
- Name two precursor lesions of SCC
- What is the name for a BCC precursor?
- Give five risk factors for NMSC
- Are KA capable of metastasis?
- In what way are IECs said to be like psoriasis?
- Name an inherited syndrome that shows an increased incidence of BCC
- Are BCC derived from the basal cell layer?
- What is it about pale skin that increases the risk of skin cancer?
(answers are found at the end of the book)